
Back |
Interactive 3D PDF of Carnegie Stage 12 |
The interactive PDF below has been very kindly provided by Dr. Jill PJM Hikspoors, Prof. Wouter H Lamers,
Department of Anatomy & Embryology, Maastricht University, Maastricht, The Netherlands
Contact: jill.hikspoors@maastrichtuniversity.nl
Publication: 10.1038/s42003-022-03153-x
*Important* Although some web browsers will allow PDFs to be viewed online, the file must be downloaded and saved to a computer to enable the interactive options.
 |
 |
 |
||
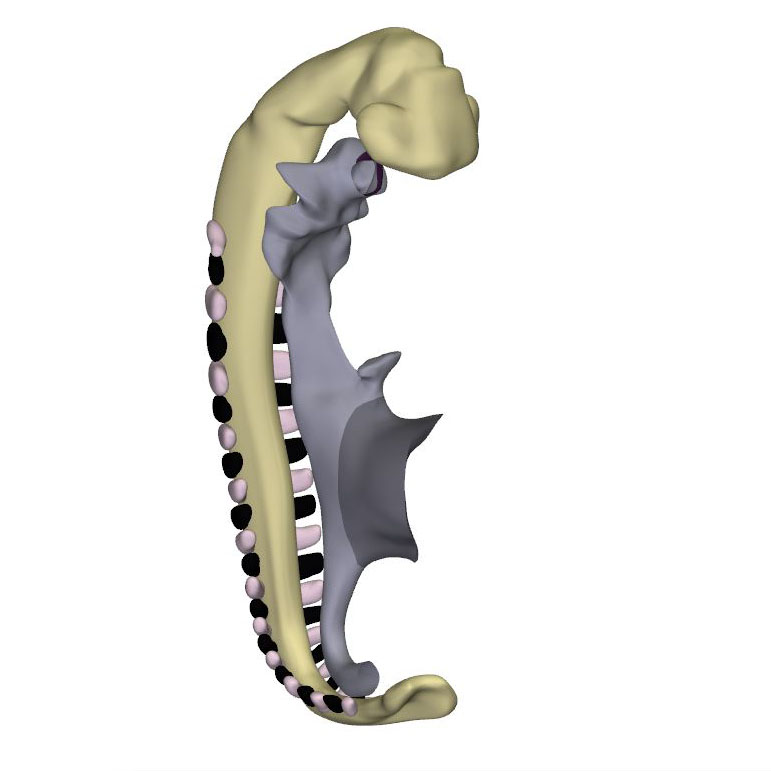
Right-sided view |
Right ventral view: Neural tube with its caudal neuropore |
Right dorsal view: Start of the inflow tract asymmetry |
||
 |
 |
 |
||
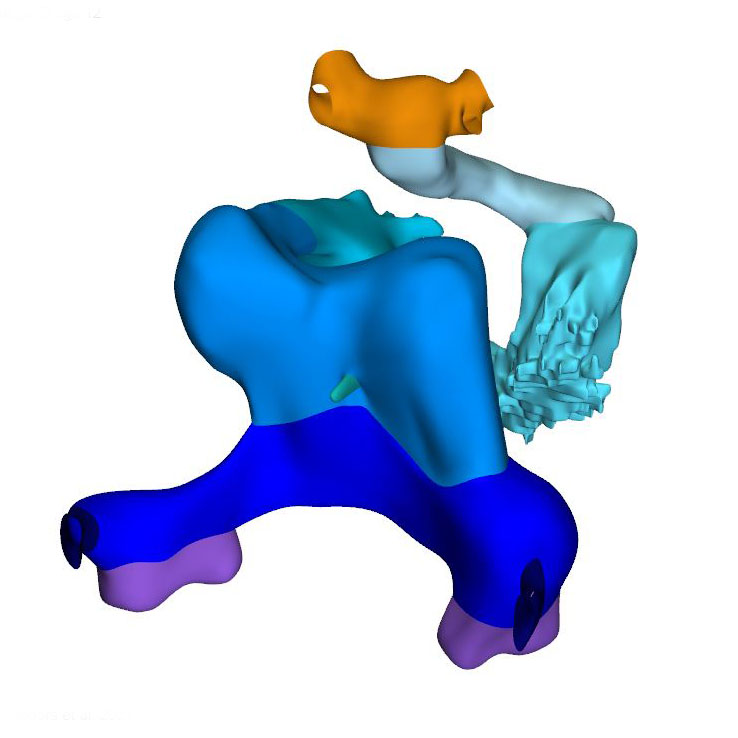
Left ventral view: Cardiac looping |
Left ventral view: Loopwire shows the cardiac looping pattern |
Caudal view: Changing position of the right ventricle |
Approximately 30 days have now passed since fertilization. The cranial neuropore has closed, while the caudal neuropore is distal to somite #23, which is equivalent to vertebral level T12. The fore- and hindgut have further elongated. The first 2 pharyngeal pouches have formed in the foregut. The buccopharyngeal membrane is breaking up. Surrounded by the pericardial cavity, the heart itself is enclosed on three sides by the transverse septum, the pharynx, and the forebrain. The large systemic veins are still bilaterally symmetrical, and common cardinal veins have formed. Already, the right-sided systemic venous sinus is expanding more rapidly than its left-sided counterpart. The bilateral proepicardial organs, which originated just cranial and lateral to the inflow tract, have expanded medially over the cranial surface of the transverse septum. A still blind mid-pharyngeal strand, which will eventually canalize to form the common pulmonary vein, can be recognized penetrating the dorsal mesocardium between the arms of the inflow tract. The atrial margin of the mesocardium is now flanked by paired mesenchymal ridges. Growth of the right-sided ridge, and its fusion with the primary atrial septum, will eventually commit the common pulmonary vein to the cavity of the left atrium. The atrioventricular canal has become recognizable, connecting the left side of the atrial components with the embryonic left ventricle.
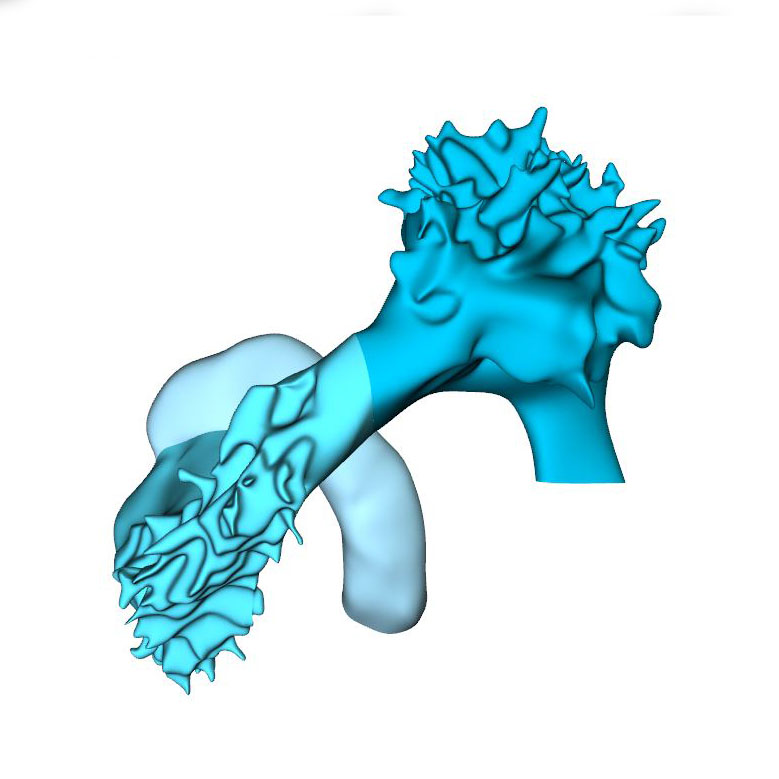
Axial growth increases the length of the heart loop, placing the ventricles in a ventral position relative to the atria, whereas radial growth results in the ballooning of the atrial appendages and apical ventricular components. The ballooning was first seen in the right atrium and left ventricle of CS11 embryos. It results from localized increases in cell proliferation in the outer curvature of the heart loop. The primary myocardium of the embryonic heart is typically bilayered, with a network of thin myocardial strands connecting the layers. The spikes that decorate reconstructions of the lumen of the embryonic left and right ventricles arise from the muscular trabeculations that appear in the ballooning portions of the ventricles between CS12 and CS15. In the embryonic left ventricle, trabeculation of the myocardial wall starts with a few endocardial sprouts penetrating the jelly at CS11. The sprouts increase in number and extend into the inner layer of the bilayered primary myocardium at CS12. At CS13, the sprouts spread laterally between both myocardial layers, inducing rearrangement of the inner myocardial layer into radial trabeculations. These muscular columns, temporarily covered by “bubbles” of jelly-like extracellular matrix, expand radially during CS14. Their resorption terminates trabecular growth at CS15. The outward and radial growth of the trabeculations leaves intact the contours of the original ventricular endocardial tube.
The appearance of endocardial sprouting into the cardiac jelly marks the morphological formation of the embryonic right ventricle at the apex of the heart loop, and in a dorsal position relative to the left ventricle. The endocardial sprouts also demarcate the left and right boundaries of the interventricular foramen. The size of the cavity, and its trabecular development, are delayed by ~2 Carnegie stages (~4 days) in the embryonic right relative to the embryonic left ventricle. Differential expression of transcription factors, including Hand1 and -2, and Tbx1 and -5, sustains the differences in growth and shape of the left and right ventricles. The embryonic right ventricle continues distally into the smooth-walled myocardial outflow tract. Separate left and right bloodstreams now already become visible in the outflow tract. Since the inner curvature of the heart does not participate in ballooning and trabeculation, the boundaries of the respective cardiac compartments can only be distinguished along the outer curvature.
When the second heart field starts to contribute cells to the arterial pole of the heart, the walls of the loop take a helical path between the atrioventricular canal and distal outflow tract. This helical configuration can be shown by the expression pattern of the left-sided marker Pitx2c, by lineage tracing and by the course of the endocardial ridges formed in the outflow tract. The spiraling direction is clockwise when looking in downstream direction. Cells from the left side of the caudal second heart field end up cranially (superiorly) in the left atrium and atrioventricular canal, whereas those from the right side end up caudally (inferiorly). Similarly, the parietal and septal ridges spiral from the right and left side of the proximal myocardial outflow tract to the left and right side of the distal myocardial outflow tract, respectively. The walls of other structures that form loops, such as the intestines, follow strikingly similar courses. During this phase of looping, the elongating muscular outflow tract forms an acute bend between its transversely oriented proximal part, which is also known as the “conus”, and its ventrodorsally oriented distal part, also known as the “truncus”. The pronounced “bayonet” or “dog-leg” bend between these parts marks the junction. This bend may be a critical structural element for effective valveless pumping in these early hearts. The presence of the bend permits the outflow tract to be described as having proximal and middle parts, which are myocardial, with the non-myocardial distal part being added when the arterial trunks begin to form in CS15 embryos (Table 1).
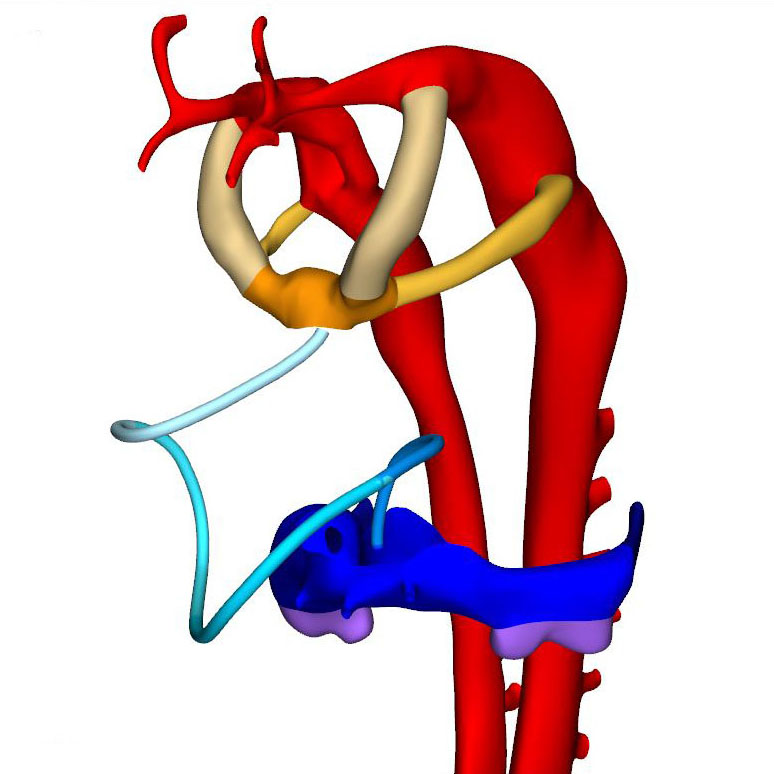
By this stage, it is possible to recognize the first two pharyngeal arches, along with their accompanying arteries. The vascular space within the ventral pharyngeal mesenchyme that connects the outflow tract with the arteries of the pharyngeal arches is known as the aortic sac. In mice, the endothelium of the first two pharyngeal arches shares its lineage with that of the dorsal and ventral aortas, but differs from that of the subsequent pharyngeal arch arteries. The dorsal aortas have fused between somites #10 and #13 (vertebral levels C6-T2), and continue cranially into the carotid arteries.
Click an image to download a 3D-PDF. The file must be saved to a computer to enable the interactive options. The 3D-PDFs can be opened on any computer as long as Adobe PDF or equivalent reader is installed.
A 3D-PDF becomes activated by “clicking” with the mouse on the reconstruction.
A toolbar appears at the top of the screen that includes the option “model tree”.
The model tree displays a material list of structures in the upper box, and preset viewing options (cameras) in the lower box. The sequence of items corresponds to that in Supplemental Table 3 of the publication.
The list of visible structures can be modified by marking or unmarking a structure.
To manipulate the reconstruction, press the left mouse button to rotate it, the scroll button to zoom in or out, and the left and right mouse buttons simultaneously to move the embryo across the screen.
A structure can be rendered transparent by selecting that option from the drop-down menu after selecting the structure with the right mouse button.
To inspect a combination of structures, one is advised to build up the composition, beginning with a familiar component, such as a lumen, rather than deleting non-relevant structures one-by-one from a completely reconstructed specimen.
The slicer button in the toolbar allows making cross sections. The plane of section can be adjusted with the offset and tilt options.
The “loop wires” in Supplemental Figures 3-6 of the publication, which are drawn through the center of the endocardial heart tube, emphasize the changing shape of the heart loop during CS10-13.
The side length of the scale cubes is 200 μm.
The preset views correspond to the images shown in Figures 1-10 of the publication.
Note that items that are visible in these views can be altered by marking or unmarking a structure in the model tree.