
Back |
Interactive 3D PDF of Carnegie Stage 15 |
The interactive PDF below has been very kindly provided by Dr. Jill PJM Hikspoors, Prof. Wouter H Lamers,
Department of Anatomy & Embryology, Maastricht University, Maastricht, The Netherlands
Contact: jill.hikspoors@maastrichtuniversity.nl
Publication: 10.1038/s42003-022-03153-x
*Important* Although some web browsers will allow PDFs to be viewed online, the file must be downloaded and saved to a computer to enable the interactive options.
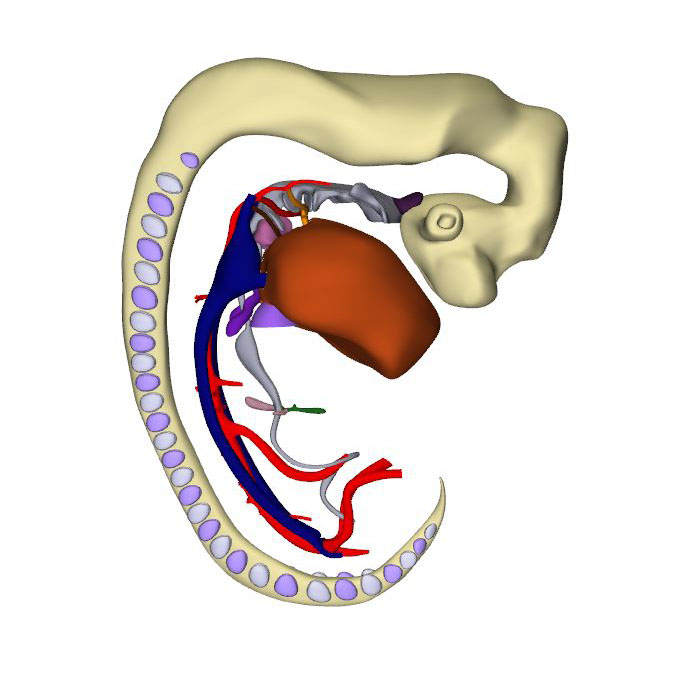 |
||||||||
Right-sided view |
Dorsal view: Note presence of left and right hepatocardiac channels |
Right sagittal view: Interatrial foramen plus secondary foramen, and interventricular foramen |
Right view: most advanced stage in which pharyngeal arch arteries retain their symmetry |
Left cranial view: GlN-positive ring surrounds interventricular foramen with atrioventricular canal (left) and outflow tract (right) |
Cranial view: Myocardial outflow tract with eliminated dog-leg bend between proximal and distal parts |
Left cranial view: aorto-pulmonary septum between aortic and pulmonary portions |
Left cranial view: prongs of neural crest cells in septal and parietal ridges |
Left cranial view: aortic and pulmonary columns and swellings occupy space in between ridges |
At this stage ~36 days have passed since fertilization. The reconstructed specimen, although one of the best CS15 specimens of this stage in the Carnegie collection, suffered from venous congestion. To decide whether or not this condition was atypical, we surveyed the Blechschmidt collection of human embryos. Eight of the 32 embryos in this collection that had been graded as good or excellent suffered from a moderate to severe degree of venous congestion. This finding suggests that cardiac failure is a common cause of death in human embryos in the 5th and 6th weeks of their development.
Compared to the embryo shown for CS14, the changes in the arrangement of the systemic veins and venous sinus are limited, except that a small left hepatocardiac channel was still present in this relatively less advanced CS15 embryo. The left hepatocardiac channel was still discernable in all four CS14 embryos, and in one of the six CS15 embryos of the Blechschmidt collection. The latter embryo did not suffer from venous congestion. The configuration of the atrial chambers and their topographic relation with the pulmonary vein are similar to those in the previous stage. The breakdown of the dorsal portion of the primary atrial septum has created, however, a secondary atrial foramen. In the Blechschmidt collection we found two CS15 embryos who had not yet formed a secondary foramen and two others who had, like the reconstructed specimen, both primary and secondary foramens. Accordingly, half of the CS15 embryos in the Carnegie collection had developed a secondary foramen. We found no notable changes in the architecture of the ventricles. The interventricular ring is still a planar structure, but a widening of the crest of the muscular ventricular septum identifies the developing branching component of the atrioventricular conduction axis.
CS15 is the most advanced stage in which the arteries within the pharyngeal arches retain their symmetry, albeit that the portions of both dorsal aortas between the arteries of the 3rd and 4th arches, known as the carotid ducts, have markedly decreased in diameter. By this stage, the arteries of the left and right 6th pharyngeal arches have each given rise to a pulmonary artery, which extends caudally within the pharyngeal mesenchyme along the trachea. The most pronounced developmental changes are to be seen in the arrangement of the middle portion of the outflow tract and the aortic sac. Continued axial growth within the myocardial part of the outflow tract has all but eliminated the dog-leg bend. The aortopulmonary septum, initially seen at CS14 as a transverse protrusion extending from the dorsal wall of the aortic sac between the origins of the arteries of the 4th and 6th pair of pharyngeal arches, now extends obliquely in a ventral direction towards the distal margins of the endocardial ridges in the middle portion of the outflow tract. In consequence, the aortic sac acquires a dextrocranial systemic component, which connects the subaortic part of the outflow tract with the arteries of the 3rd and 4th pharyngeal arches, and a sinistrocaudal pulmonary component, which connects the subpulmonary part of the outflow tract with the arteries of the 6th arches. The intrapericardial part of the systemic component can be labeled with Mef2c-Cre, an often-used marker of the cranial second heart field, and becomes the ascending aorta. The lateral horns of the aortic sac remain unlabeled, and become the extrapericardial part of the ascending aorta, the brachiocephalic trunk, and the initial part of the transverse aortic arch. The pulmonary component becomes the pulmonary trunk, an entirely intrapericardial vessel. It is the ventral growth of the aortopulmonary septum, therefore, which initiates the anatomical separation of the arterial pole of the heart, along with the formation of the non-myocardial distal portion of the outflow tract. The configuration of the distal myocardial jaws and the interposed mural columns remains unchanged relative to that in CS14. Accordingly, the distal myocardial jaws still extend close to the pericardial reflection. As in the CS14 embryos, tissue with the phenotypic property of the pulmonary mural column extends dorsally to the dense mesenchyme that surrounds the trachea.
The prongs of neural crest cells, which dorsally are continuous with the neural crest cells in the pharyngeal floor and the aortopulmonary septum, can now be traced ventrally into the proximal outflow ridges. By this stage, it becomes possible to recognize the sites of formation of the arterial valves as increasingly narrow passages between the endocardial ridges medially and the swellings laterally. These passages are recognizable histologically by their lining with intensely staining, cobble stone-shaped endocardium. Separate aortic and pulmonary channels are now identifiable in luminal casts of the middle portion of the outflow tract. They extend from the initial site of the dog-leg bend to the distal boundary of the myocardium of the outflow tract. Until fusion of both endocardial ridges occurs during CS17, the subaortic and subpulmonary channels remain connected by an aortopulmonary foramen, which is bounded dorsally by the leading edge of the aortopulmonary septum. At this distal location the parietal and septal ridges occupy craniosinistral and caudodextral positions, respectively, with the still small swellings occupying the spaces in between.
Click an image to download a 3D-PDF. The file must be saved to a computer to enable the interactive options. The 3D-PDFs can be opened on any computer as long as Adobe PDF or equivalent reader is installed.
A 3D-PDF becomes activated by “clicking” with the mouse on the reconstruction.
A toolbar appears at the top of the screen that includes the option “model tree”.
The model tree displays a material list of structures in the upper box, and preset viewing options (cameras) in the lower box. The sequence of items corresponds to that in Supplemental Table 3 of the publication.
The list of visible structures can be modified by marking or unmarking a structure.
To manipulate the reconstruction, press the left mouse button to rotate it, the scroll button to zoom in or out, and the left and right mouse buttons simultaneously to move the embryo across the screen.
A structure can be rendered transparent by selecting that option from the drop-down menu after selecting the structure with the right mouse button.
To inspect a combination of structures, one is advised to build up the composition, beginning with a familiar component, such as a lumen, rather than deleting non-relevant structures one-by-one from a completely reconstructed specimen.
The slicer button in the toolbar allows making cross sections. The plane of section can be adjusted with the offset and tilt options.
The “loop wires” in Supplemental Figures 3-6 of the publication, which are drawn through the center of the endocardial heart tube, emphasize the changing shape of the heart loop during CS10-13.
The side length of the scale cubes is 200 μm.
The preset views correspond to the images shown in Figures 1-10 of the publication.
Note that items that are visible in these views can be altered by marking or unmarking a structure in the model tree.