
Back |
Interactive 3D PDF of Carnegie Stage 23 |
The interactive PDF below has been very kindly provided by Dr. Jill PJM Hikspoors, Prof. Wouter H Lamers,
Department of Anatomy & Embryology, Maastricht University, Maastricht, The Netherlands
Contact: jill.hikspoors@maastrichtuniversity.nl
Publication: 10.1038/s42003-022-03153-x
*Important* Although some web browsers will allow PDFs to be viewed online, the file must be downloaded and saved to a computer to enable the interactive options.
 |
 |
 |
 |
|||
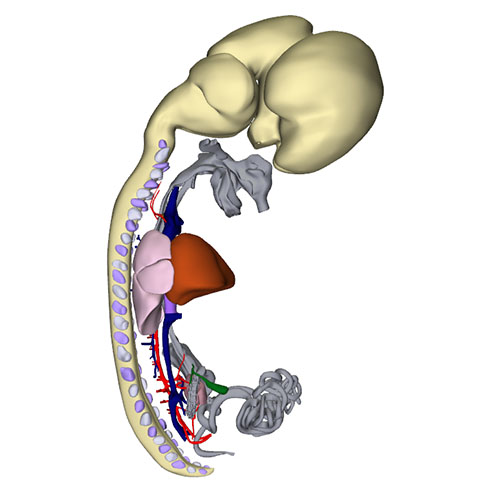
Right-sided view |
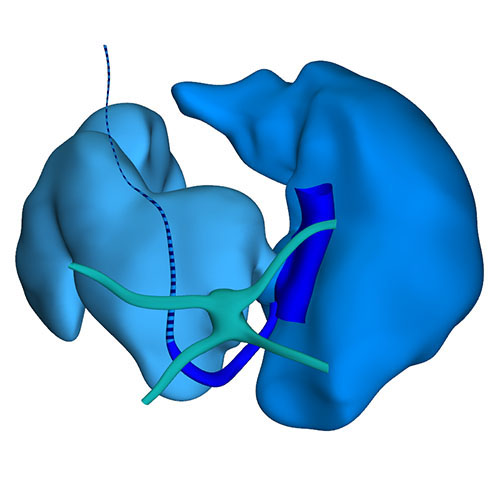
Dorsal view: Incorporation of the stem pulmonary vein and remaining part of the left sinus horn (vein of Marshall) |
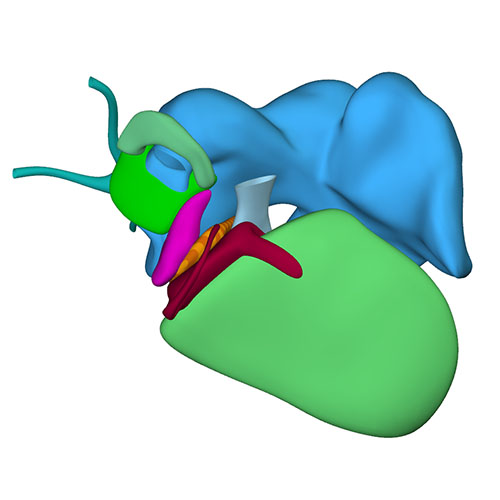
Right sagittal view: Visualizing secondary atrial septum |
Left cranial view: myocardialized aortopulmonary septum |
|||
 |
 |
 |
||
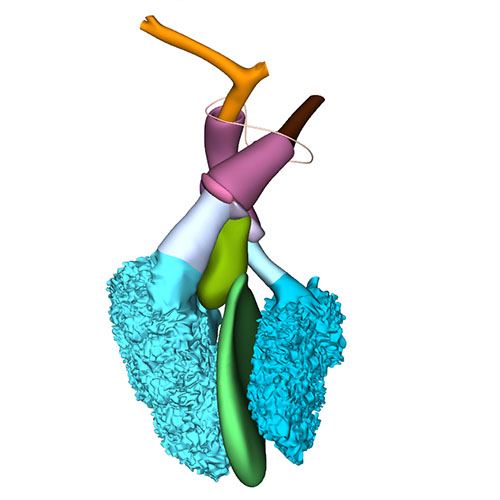
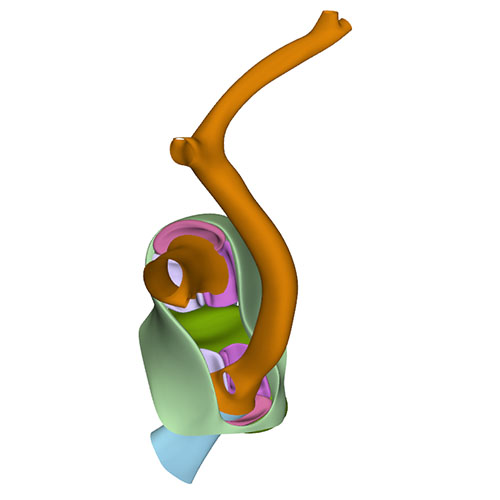
Left cranial view: Semilunar valves and wall arterial trunks |
Dorsal view: Semilunar valves and asymmetric aortic trunk |
Ventral view: Changing position of the right ventricle |
At this last Carnegie stage, ~56 days have elapsed since fertilization. Between CS14 and CS23 the diameter of the left sinus horn does not change, implying that it receives an increasingly smaller percentage of the systemic venous blood. Accordingly, the left cardinal vein starts to attenuate between the junction of the left subclavian and jugular veins cranially, and its passage in the left atrial ridge between the left inferior pulmonary vein and the left atrial appendage caudally. Meanwhile, the brachiocephalic vein is forming from merging venous spaces that arise between both jugular veins just cranial to the aortic arch. Remodeling occurs between CS20 and CS21, with only a minute left common cardinal vein present at CS22, the lumen of which has disappeared at CS23. The distal obliterated part of the left sinus horn is known as the ligament of Marshall, whereas the remaining proximal part is known as the coronary sinus. The atrial appendages have increased further in size, and their pectinate muscles are well developed. Myocardium now surrounds the pulmonary veins up to their second division. The diameter of the stem of the pulmonary veins continues to increase, preluding the incorporation of the pulmonary veins as 2 separate tributaries into the roof of the left atrium in the 9th week of development and as 4 tributaries in the 14th week. A fold now begins to form in the roof of the right atrium just rightward of the primary atrial septum. It is against this fold, which is incorrectly known as the secondary atrial “septum”, that the primary septum will eventually rest to close the oval foramen.
By now, the leaflets of the atrioventricular valves are forming, although tendinous cords have yet to develop. The leaflets, furthermore, still contain myocardium on their ventricular surface. Fragmentation of the myocardial floor of the tricuspid gully gives atrial blood access to the right ventricular cavity via conduits that pass the septomarginal trabeculation cranially (pre-existing) and caudally (newly formed). In both ventricles, the papillary muscles begin to form by consolidation of aggregating trabeculations, with the compaction starting at the valve leaflets and moving in the direction of the compact ventricular walls. Epicard-derived cells have begun to induce insulation within the atrioventricular junctions, and have populated the lateral cushions. During CS21 and CS22, the whorl of neural crest cells in the ridges of the proximal outflow tract all but disappears, while myocardialization continues. As already explained, the so-called tendon of the conus, a cord-like band between the aortic and pulmonary roots, is an inconsistent distal remnant of the whorl. The still long subaortic outlet now passes between the developing mitral valve, the muscular ventricular septum, and the muscularized septum in the proximal outflow tract. The subpulmonary outlet passes between the muscularized septum in the proximal outflow tract and the free right ventricular wall.
The muscular septum in the proximal outflow tract, also known as outlet septum, is normally a temporary embryological structure. It changes in shape and orientation from a dumbbell-like structure perpendicular to the muscular ventricular septum at CS20 to a flat blade almost parallel to the muscular ventricular septum at CS23. Extension of its myocardialization towards the developing arterial roots underlies this change in orientation. This positional change coincides with the incorporation of the proximal outflow tract into the ventricles, and transforms the transitory outlet septum into the smooth medial wall of the right ventricle, the dorsocranial part of which becomes the “free-standing” muscular subpulmonary infundibulum. The attribution of the muscular septum of the outflow tract as a mostly right-ventricular structure can be best appreciated if the ventricular cavities, muscular septum, and (sub-)aortic and (sub-)pulmonary channels are observed from the left. The developmental events underlying the transformation of the embryonic outlet septum from a septal to a mural structure are still poorly understood, but probably reflect the asymmetric growth of the increasingly wedge-shaped and transversely oriented right ventricle. Should the middle portion of the interventricular foramen fail to close, then the result will be a perimembranous ventricular septal defect. Should the asymmetric growth of the right ventricle and the transfer of the aortic root to the left ventricle be hampered, however, the result will be tetralogy of Fallot, or double-outlet right ventricle. In all these settings, it remains possible to recognize a muscular or fibrous outlet septum.
Between CS18 and CS23, the arterial valvar leaflets become slenderer, the walls of the sinuses better formed, and the ventriculoarterial and sinutubular junctions identifiable structures. As development progresses, the myocardial cells of the valvar cuff covering the left and right leaflets do not proliferate, but become diluted in the surrounding proliferating epicardial connective tissues. Remnants of this myocardium can, nevertheless, persist at least until the 3rd trimester (our unpublished observations) and perhaps into adult life.
Click an image to download a 3D-PDF. The file must be saved to a computer to enable the interactive options. The 3D-PDFs can be opened on any computer as long as Adobe PDF or equivalent reader is installed.
A 3D-PDF becomes activated by “clicking” with the mouse on the reconstruction.
A toolbar appears at the top of the screen that includes the option “model tree”.
The model tree displays a material list of structures in the upper box, and preset viewing options (cameras) in the lower box. The sequence of items corresponds to that in Supplemental Table 3 of the publication.
The list of visible structures can be modified by marking or unmarking a structure.
To manipulate the reconstruction, press the left mouse button to rotate it, the scroll button to zoom in or out, and the left and right mouse buttons simultaneously to move the embryo across the screen.
A structure can be rendered transparent by selecting that option from the drop-down menu after selecting the structure with the right mouse button.
To inspect a combination of structures, one is advised to build up the composition, beginning with a familiar component, such as a lumen, rather than deleting non-relevant structures one-by-one from a completely reconstructed specimen.
The slicer button in the toolbar allows making cross sections. The plane of section can be adjusted with the offset and tilt options.
The “loop wires” in Supplemental Figures 3-6 of the publication, which are drawn through the center of the endocardial heart tube, emphasize the changing shape of the heart loop during CS10-13.
The side length of the scale cubes is 200 μm.
The preset views correspond to the images shown in Figures 1-10 of the publication.
Note that items that are visible in these views can be altered by marking or unmarking a structure in the model tree.